Part One: The Known
When you think of what makes up the universe, you will probably think of atoms. Sure, they are the building blocks to most things we see around us, but as we discovered in a previous post, atoms aren’t the end of the story. The atoms are made up of smaller particles described by the Standard Model, which not only includes types of quark (which make up protons and neutrons) and leptons but also bosons (force-carrying particles). Then surely the rest of the universe is made up of all of them? Its true, the universe is filled with electromagnetic waves (photons) and atoms. When we look out into the galaxy and beyond, we see an abundance of elements such as Hydrogen, Helium and Carbon, and for decades scientists thought that was it. But it turns out… all the things that we can see, everything we can observe, makes up a measly 4% of the universe!
What do we know?
We know that there are 118 elements, 96 of them naturally occurring as a result of nuclear fusion in stars, or the intense heat and pressure from a supernova. These elements took millenia to discover and understand, and the periodic table took 150 years to complete.

The most abundant elements in the universe are Hydrogen (the first element produced after the Big Bang), Helium (produced seconds after Hydrogen from high-speed collisions) and then Oxygen and Carbon. The reason the next most common elements aren’t the next lightest (Lithium and Beryllium) is that Lithium is rarely formed in stars and despite being made just after Helium, it was a comparatively small amount, and Beryllium often doesn’t survive supernovae, and is instead used to create heavier elements. Other than this, element abundance mainly falls with increasing atomic mass.
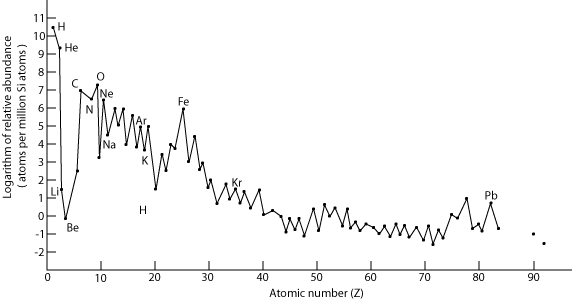
From: http://www.algebralab.org/img/5ee9a9c6-32b5-4762-8e34-517122e67f92.gif
Just a point of clarification: only the nuclei of Hydrogen, Beryllium and Lithium were created seconds after the big bang; it took 380,000 years for the universe to cool to a temperature at which electrons could be held in place around them.
We know that these elements are further split into quarks and electrons, which are only a small part of a group of matter particles, called fermions, of which there are twelve: there are 6 quarks (3 positive and 3 negative) which interact with the Strong Force, and 6 Leptons (3 negative, 3 neutral) which don’t. If you want to find about more about The Standard Model, go to my Particle Physics page
The other half of the Standard Model describes Bosons. These bosons ‘carry’ different forces that allow interactions between particles to take place.
The main Bosons (called Gauge Bosons) are:
- Photons: carry the Electromagnetic Force (you’ve probably heard of these)
- This is what keeps particles that are of opposite charges attracted, for example electrons to the nucleus. It is also for this that the opposite is true: like charges repel.
- They also carry electromagnetic waves, which we see as light, which are produced from a change in energy of a particle.
- Gluons: carry the Strong Force
- It is what keeps quarks binded, and as a result is what binds the protons and neutrons in an atomic nucleus together. This is arguably the most important of the 4, as its primary purpose within an atom is to overcome the strength of the Electromagnetic Force that would otherwise push apart protons.
- W bosons: carry the Weak Force
- Changes a particle’s inherent charge, which actually means the particle changes to another (for example Up-type Quark into a Down-type Quark)
- Therefore both they and Z Bosons (see below) play an important role in the decay of atoms.
- Z Bosons: also carry the Weak Force
- Can change a particle’s energy but not its charge
There is also the Higgs Boson, a relatively recent discovery, which I will cover in a future post.
These particles all coalesce into the world that we live in, and for most of human history, we thought that was it!

So, Standard Matter- everything you can see, observe and/or describe and their constituents- is pretty well understood to a certain extent, as it generally interacts normally/predictably with other matter and energy. The only uncertainty comes from the smallest particles, as quantum mechanics is rather odd, but, we still have good equations that describes it!
The rest, due the stretch of modern technology, is basically invisible. This huge chunk (96%) of our universe has been split into two, unimaginatively named, sections: dark matter, and dark energy.
Check back tomorrow for Part 2, where I’ll explore why Dark Energy and Dark Matter must be there, and what they could possibly be made of!
☆☆☆
If you can’t trust an atom… trust in science!
☆it’s like magic, but it’s true whether you believe in it or not!☆
See you next time!
☆☆☆



woh I like your posts, bookmarked! .
LikeLiked by 1 person
Thank you so much!
LikeLike